|
Home runs travel farther in the less dense air in high-altitude Denver than in lower-altitude ballparks, according to baseball players. The term for this type of air resistance is "drag," and it increases as air density rises. Objects traveling through denser, or "heavier," air will slow down more because the object must, in effect, push away more or heavier molecules. 
Therefore, the new density of the air becomes doubled when the pressure of the air is doubled keeping the temperature constant. Therefore, density of air is directly proportional to the pressure of the air.So, when the the pressure of the air is doubled keeping the temperature constant, then the new density of air is given by,ĭ’ - the new density of air when pressure is doubled at constant temperature. Sol: We know the relationship between density of air and pressure given by the formula, What is the density of air when the pressure of the air is doubled keeping the temperature constant? Substitute the equation for number of moles ( n ) in the ideal gas equation(1) and simplify The formula to calculate the number of moles is given by , To derive the density of air formula, we use the ideal gas equation given by So they need oxygen cylinders for easy breathing. That is why mountaineers carry oxygen cylinders because the density of air is less at higher altitudes. So the density of air is maximum at sea level. As we are moving up away from the sea level, the density of air decreases. Variation of Density of Air at Different AltitudesĪs we know the density of air in the atmosphere changes its value from place to place. Consider an air taken in a container of volume 1 m 3, then the mass of the air inside that container having volume 1 m 3 is equal to the density of air.There are some factors that the density of air depends upon. The S.I unit of density of air is kg/m 3. The density of air is defined as the mass per unit volume of air. The majority of first-year chemistry textbooks describe how this works. In layman's terms, he discovered that no matter what gas is in the container, a fixed volume of gas, say one cubic metre, at the same temperature and pressure, will always have the same number of molecules. Lorenzo Romano Amedeo Carlo Avogadro (9 August 1776 – 9 July 1856) was an Italian scientist best known for his contribution to molecular theory, which is now known as Avogadro's law, which states that equal volumes of gases at the same temperature and pressure contain equal numbers of molecules.Īmadeo Avogadro, discovered it in the early 1800s. 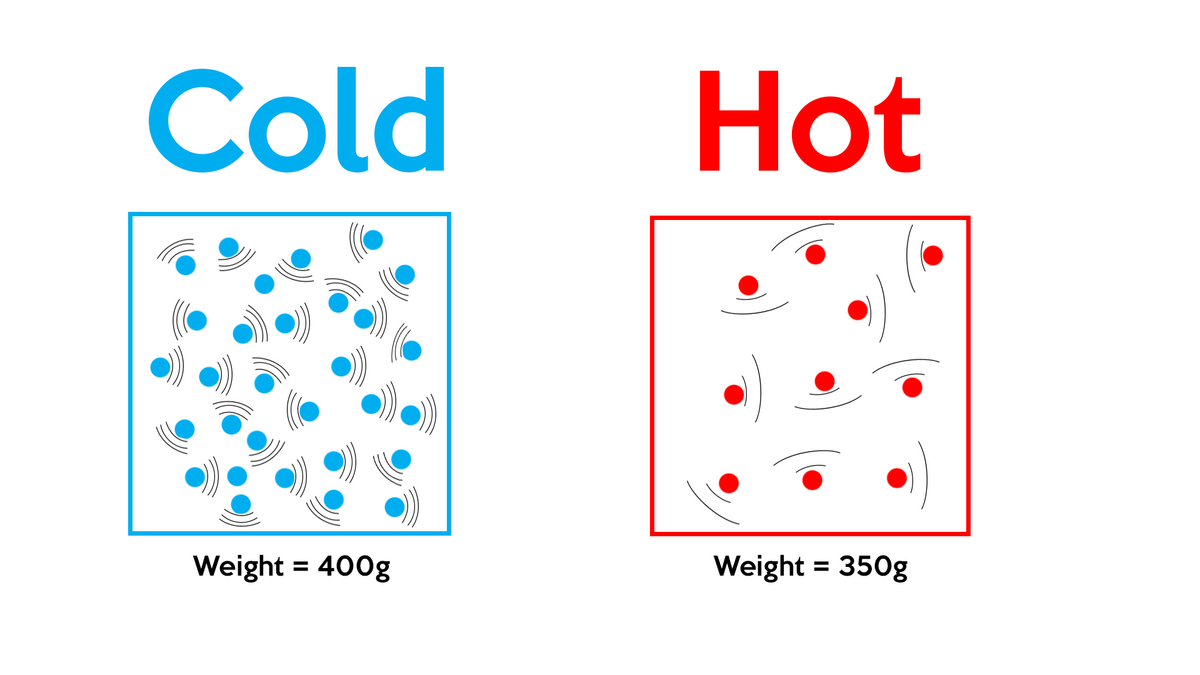
Here, we will discuss how to calculate the value of air density at different temperatures and pressures using the density of air formula. In the case of the atmosphere, the density of air varies from place to place. There are some factors that the density of the air depends upon. Therefore, density of air is the mass of air contained in a unit volume of a container. The density of a body is the mass of the body per unit volume.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
